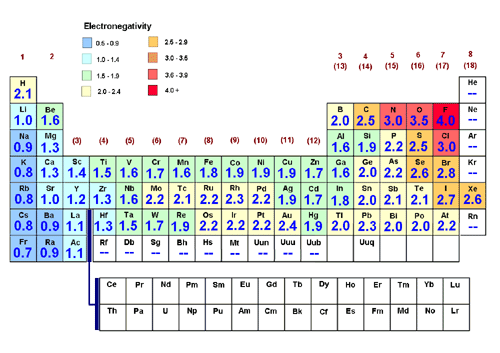
Electronegativity:
Electronegativity is the measure of the ability of an atom in a chemical compound to attract electrons from another compound. The most electronegative element is fluorine(F). Its value is 4.0. All other elements are compared and based off of. Electronegativity has patterns or trends across the periods and the groups. The basic trend for electronegativity across periods is from left to right the electronegativity value tends to increase. This occurs because of the nuclear charge. Going from left to right across a period, the elements gain electrons or nuclear charge. This nuclear charge allows the atom to attract electrons thus making it more electronegative. The less amount of protons leads to less attraction between electrons and protons. The trend for electronegativity down a group starting from top to bottom is a decrease in electronegativity value. This occurs because the atomic radius gets larger to make room for the electrons, this creates less attraction between the the electrons and protons making it harder for the atom to attract electrons. Another reason for this trend is the shielding effect which limits attraction, and the atom’s ability to attract electrons.
Electronegativity is the measure of the ability of an atom in a chemical compound to attract electrons from another compound. The most electronegative element is fluorine(F). Its value is 4.0. All other elements are compared and based off of. Electronegativity has patterns or trends across the periods and the groups. The basic trend for electronegativity across periods is from left to right the electronegativity value tends to increase. This occurs because of the nuclear charge. Going from left to right across a period, the elements gain electrons or nuclear charge. This nuclear charge allows the atom to attract electrons thus making it more electronegative. The less amount of protons leads to less attraction between electrons and protons. The trend for electronegativity down a group starting from top to bottom is a decrease in electronegativity value. This occurs because the atomic radius gets larger to make room for the electrons, this creates less attraction between the the electrons and protons making it harder for the atom to attract electrons. Another reason for this trend is the shielding effect which limits attraction, and the atom’s ability to attract electrons.