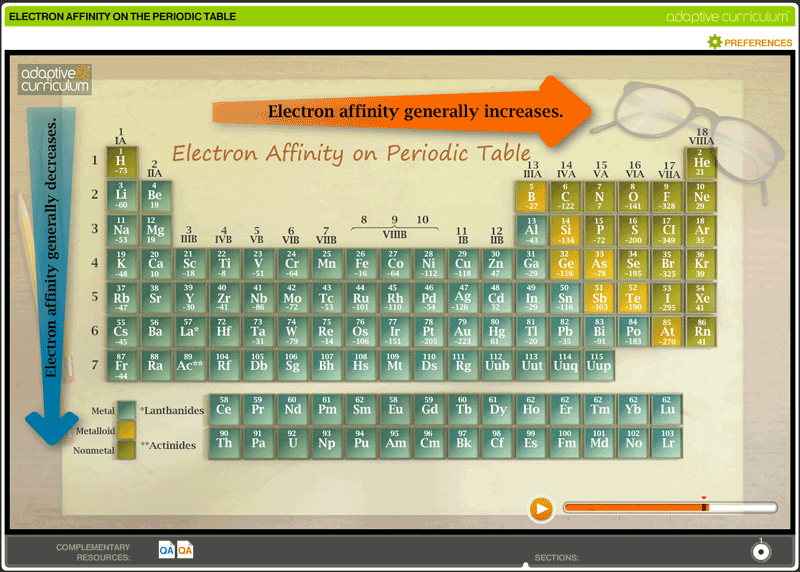
Electron Affinity:
Electron affinity is the energy change that occurs when an electron is added to a neutral atom. Electron affinity is rarely applied to second or third electron affinity levels because the elements are so unstable, and thus unable to survive long in nature. If they do exist the values for the electron affinities are all positive. It is measured in kJ/mol. Electron affinity also experiences trends or patterns in both periods and groups. The electron affinity trend across periods from left to right is from left to right, the electron affinity increases in electron affinity value although the numbers become less and less(negative values). A prime example of electron affinity is Group 17: halogens. The halogens are a group of elements who share the property of needing a single electron to become stable. This property makes the halogens ready and happy to bond with other atoms to fill the last space in order to achieve noble gas configuration. The group trends for electron affinity is from top to bottom the electron affinity decreases. It takes more energy to add electrons the farther down the group is. The reason for this is nuclear charge which causes increase in electron affinities. Another reason for the electron affinity group trend is increased in atomic radius. The increase in the atomic radius lessens attraction between the nucleus and other electrons thus making it harder for electrons to join an atom if they are not attracted to it.
Electron affinity is the energy change that occurs when an electron is added to a neutral atom. Electron affinity is rarely applied to second or third electron affinity levels because the elements are so unstable, and thus unable to survive long in nature. If they do exist the values for the electron affinities are all positive. It is measured in kJ/mol. Electron affinity also experiences trends or patterns in both periods and groups. The electron affinity trend across periods from left to right is from left to right, the electron affinity increases in electron affinity value although the numbers become less and less(negative values). A prime example of electron affinity is Group 17: halogens. The halogens are a group of elements who share the property of needing a single electron to become stable. This property makes the halogens ready and happy to bond with other atoms to fill the last space in order to achieve noble gas configuration. The group trends for electron affinity is from top to bottom the electron affinity decreases. It takes more energy to add electrons the farther down the group is. The reason for this is nuclear charge which causes increase in electron affinities. Another reason for the electron affinity group trend is increased in atomic radius. The increase in the atomic radius lessens attraction between the nucleus and other electrons thus making it harder for electrons to join an atom if they are not attracted to it.